
Longread
Assessing the quality of fruits by monitoring Volatile Organic Compounds
Have you ever smelled melons in the supermarket to ensure that you pick a mature one? Or have you ever been struck by the harsh smell of rotten potatoes? Attractive or repulsive odours are one of the strategies used by plants to assure their defence or propagation. One of the most important quality aspects of fruits and vegetables is their aroma. At the origin of both the aroma and flavour of food products are the volatile organic compounds (VOCs).
Volatile organic compounds
In fresh fruits and vegetables, VOCs are relatively small molecules that can be released at room temperature. VOCs are constantly released by food products at all stages of the food-production chain “from farm to fork” and they are key drivers of food perceived quality both before (odour), during (flavour and aroma) and after (aftertaste, after-flavour) food consumption. Measuring VOCs released by fresh products like fruits and vegetables (F&V) can help in evaluating their sensory qualities and can also provide information regarding their origin and traceability. Moreover, since fresh F&V are living products that age, measuring the VOCs released by respiration or senescence processes allow us to collect information about the products’ metabolic status including ripeness, senescence and the presence of infections by pathogens.
In postharvest stress monitoring, so-called ‘biotic’ and ‘abiotic’ stress is distinguished. VOCs released due to biotic stress are generally related to a bacterial or fungus infection. F&V respond to microbial infection by releasing VOCs as stress indicators for their environment, digestion products of the F&V produced by the microbiota and the VOCs produced by the microbiota itself. Detecting a microbial infection in an early stage is mostly of utmost importance to respond early and to prevent product loss. Abiotic stress refers to physical factors that can influence the F&V quality, for example, storage temperature, light, relative humidity, hydration or drought and the concentration of oxygen and carbon dioxide during storage.
Especially for biotic stress, monitoring volatile production during fresh product storage has become increasingly important in the postharvest domain. Measuring VOCs is appealing because in most cases they can be performed non-invasively and non-destructively meaning that no F&V products are lost in the process.
VOCs in postharvest
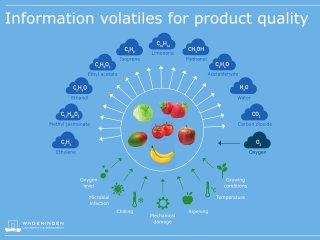
More than 12,000 flavour compounds have been reported in various foods and it has been estimated that 600-1200 (5-10%) of those compounds play a significant role in the formation of specific aromas VOCs for food products. For F&V not only flavour compounds but also non-aromatic VOCs are of interest. These non-aromatic VOCs are monitored during the storage of fresh products. The most important volatile compounds usually monitored in the postharvest domain are:
- Air constituents like oxygen (O2) and carbon dioxide (CO2). Although O2 and CO2 are strictly speaking not volatile “organic” compounds, they are relevant during storage where for example controlled atmosphere or modified atmosphere packaging is applied. Furthermore, increased consumption of oxygen and higher emissions of carbon dioxide may indicate a high respiration rate of the products. These can be indicators of biotic and abiotic stress or tell something about the ripening processes and stage of the F&V.
- Plant hormone VOCs, like the well-known ethylene, for climacteric ripening of fruits and senescence. For each type of fruit, a cocktail of different VOCs is important for ripening and senescence; in general ethanol, acetaldehyde, and nitric oxide are associated with the ripening process, whereas methyl salicylate and methyl jasmonate are plant growth regulators which modulate the plant defense response.
- Fermentation markers: ethanol and methanol. These alcohols are often used as markers of the fermentation of fruit. Ethanol is mainly produced by F&V when the anaerobic respiration pathway is used by the product. Methanol usually derives from the degradation of the membrane’s pectin and, when present, the woody parts of the fruit.
- Bacterial and mould infection markers: other compounds such methyl acetate, ethyl acetate, acetone, and propanol can indicate infection biological processes occurring on or into the F&V. Biomarkers of pathogens infection are different VOCs such as aldehydes, ketones, acids, alcohols, esters and sulphur compounds.
Detection of VOCs in the postharvest chain
Measuring and monitoring VOCs can be an incredibly challenging task, due to their versatility, low concentration in the air, high volatility, and resemblance to each other by for example molecular structure or molecular weight. Furthermore, possible cross-contamination of the VOCs measurement can occur when packing material or storage facilities released VOCs in the direct surrounding of the product. For this reason, different analytical methods have been developed to either focus on the detection of certain VOCs or to measure a profile of all VOCs coming from the product. In the table below, an overview of the most used methods for measuring VOCs in postharvest is summarized.
| Measurement method | Device / Technology | Lowest concentration | Sampling | Used mostly in |
|---|---|---|---|---|
| Air constituents detection (O2 and CO2) | Gas chromatography - Thermal conductivity detector (GC-TCD) | Ppb | Non-destructive | Research |
| Air constituents detection (O2 and CO2) | Zirconia and IR - dual bean sensors | % | Non-destructive | Practice |
| Ethylene detection and quantification | Gas chromatography - Flame Ionisation Detector (GC-FID) | Ppb | Non-destructive | Research |
| Ethylene detection and quantification | Photo sensor | Ppb - Ppt | Non-destructive | Practice |
| Ethanol | Solid state electrochemical sensor | Ppb | Non-destructive | Practice |
| Methanol and Ethanol | e-Noses in combination with solid phase micro-extraction (SPME) | Ppb | Destructive | Research |
| Total VOC profile, Volatilome | Thermo Desorption GC-MS and Direct Ionization Mass Spectrometry | Ppb | Non-destructive | Research |
| Total VOC profile, Volatilome | Proton-transfer reaction mass spectrometry | Ppb | Non-destructive | Research |
Note: Ppb = parts per billion or 1 microgram per liter; Ppt = parts per trillion or 1 nanogram per liter.